AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Ortho meta para8/14/2023  
deactivates ring by induction, slows reaction rate Halogens can also donate electron density via a resonance effect of the lone pair of electrons. reaction sites xĪ halogen substituent -X (-F, -Cl, -Br, -I) will withdraw electron density via an inductive effect due to high electronegativity. Deactivating substituents such as carbonyl or -CF3 are therefore generally meta directors. Ortho/para attack is bad, meta attack is no problem. xħ Deactivating Substituents – meta directors Meta attack gives a delocalized carbocation without any special destabilization, so meta is favored. xĦ Deactivating Substituents – meta directorsįor deactivating sustituents, cation intermediates formed from ortho or para attack are destabilized. /ortho-meta-para-xylene-56a12db43df78cf772682c56.png)
Meta attack gives a delocalized carbocation without any special stabilization. 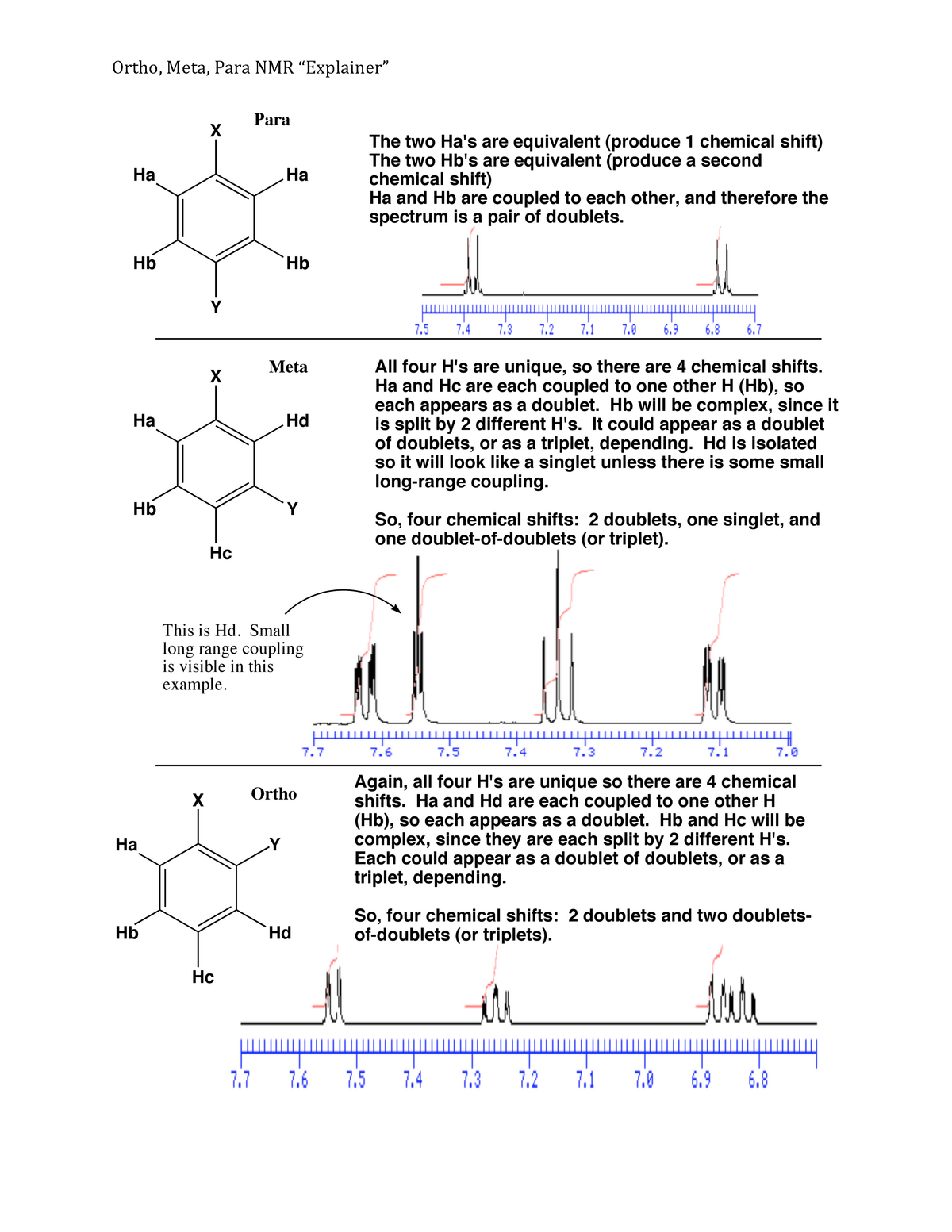
meta not stabilizedĤ Activating Substituents – o/p directorsįor activating sustituents, cation intermediates formed from ortho or para attack are stabilized, so reaction there is favored. Direct substitution to the ortho/para positions. (They also are activators.) meta not stabilizedĪlkoxyl, and amino groups have a strong, electron-donating resonance effect. For benzene + Br2 : Intermediate: in the delocalized carbocation intermediate, charge is always on ortho and para carbons The site of reaction will depend on the effect the substituent has on the carbocation intermediate.Īlkyl groups direct substitution to positions ortho and para to themselves. Examine mechanism of the substitution reaction. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
